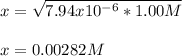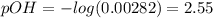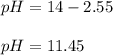Chemistry, 13.06.2021 02:30 madams1820
A 25.0 mL solution of quinine was titrated with 0.50 M hydrochloric acid, HCl. It was found that the solution contained 0.100 moles of quinine. What was the pH of the solution after 50.00 mL of the HCl solution were added

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 10:50
Someone offer some answers to this, i will give 98 coins and mark as brainliest! i will put the rest of the lab down in the comments,solutions pre-lab questions: in this lab, you will make fruit drinks with powdered drink mix. complete the pre-lab questions to get the values you need for your drink solutions. calculate the molar mass of powered fruit drink mix, made from sucrose (c12h22o11).using stoichiometry, determine the mass of powdered drink mix needed to make a 1.0 m solution of 100 ml. (hint: use molarity = to find the moles of drink mix, then convert moles to grams using a mole conversion.)what mass of powdered drink mix is needed to make a 0.5 m solution of 100 ml?
Answers: 1

Chemistry, 22.06.2019 14:20
Which statement explains why the bonds between non metals tend to be covalent? the bonds are found to be nondirectional they have large differences in electronegativity they have small differences in electronegativity they have ions that produce an electrostatic pull
Answers: 1

Chemistry, 22.06.2019 14:50
Which of the following is most likely true about water in chemical systems? a) water dissolves nonpolar ionic compounds. b) water dissociates ionic compounds. c) water dissociates covalent molecules. d) water dissolves nonpolar covalent substances.
Answers: 1

Chemistry, 22.06.2019 22:30
Which is a characteristic of the electron sea model for metallic bonding? molecular orbitals overlap to produce bands. electrons flow easily between metal nuclei. electrons are in fixed positions in the orbitals. atomic nuclei are arranged in an irregular pattern.
Answers: 3
You know the right answer?
A 25.0 mL solution of quinine was titrated with 0.50 M hydrochloric acid, HCl. It was found that the...
Questions

Mathematics, 03.09.2021 06:10


Biology, 03.09.2021 06:10

Mathematics, 03.09.2021 06:10


Computers and Technology, 03.09.2021 06:10

Mathematics, 03.09.2021 06:10


Mathematics, 03.09.2021 06:10


Mathematics, 03.09.2021 06:10


English, 03.09.2021 06:10

Mathematics, 03.09.2021 06:10

Mathematics, 03.09.2021 06:10

Mathematics, 03.09.2021 06:10

Mathematics, 03.09.2021 06:20


Mathematics, 03.09.2021 06:20

Health, 03.09.2021 06:20

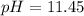
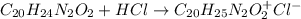
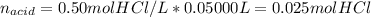
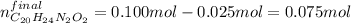
![[C_{20}H_{24}N_2O_2]=\frac{0.075mol}{(0.025+0.050)L} =1.00M](/tpl/images/1372/8041/2f907.png)
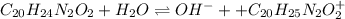
![Kb=\frac{[C_{20}H_{24}N_2O_2][C_{20}H_{25}N_2O_2 ^+]}{[C_{20}H_{24}N_2O_2]} \\\\10^{-5.1}=\frac{x^2}{1.00M}\\\\ 7.94x10^{-6}=\frac{x^2}{1.00M}](/tpl/images/1372/8041/d615c.png)