
Chemistry, 12.06.2021 16:20 laiba012305
A 5.00 mL sample of vinegar, a solution of acetic acid, is titrated with 0.756 M calcium hydroxide. 10.23 mL of the calcium hydroxide is required to reach the equivalence point. What is the molarity of the acetic acid in the vinegar.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:50
What is the overall order of reaction for rate = k[no]2[o2]
Answers: 3

Chemistry, 22.06.2019 19:00
A4.86 g piece of metal was placed in a graduated cylinder containing 15.5 ml of water. the water level rose to 17.3 ml. what is the density of the metal. i need the steps of how to solve it to so i can use a formula to work out other problems.
Answers: 1

Chemistry, 23.06.2019 11:00
Asolubility table shows that almost all compounds of group 1 metals are soluble in water. this general rule tells you that mgi2 is soluble rbno3 is soluble cacl2 is soluble co2 is soluble
Answers: 1

Chemistry, 23.06.2019 14:00
What is the final volume in milliliters when 0.641 l of a 34.0 % (m/v) solution is diluted to 23.5 % (m/v)?
Answers: 1
You know the right answer?
A 5.00 mL sample of vinegar, a solution of acetic acid, is titrated with 0.756 M calcium hydroxide....
Questions




Mathematics, 22.10.2019 06:00

Mathematics, 22.10.2019 06:00

History, 22.10.2019 06:00


Physics, 22.10.2019 06:00


Chemistry, 22.10.2019 06:00

Physics, 22.10.2019 06:00

History, 22.10.2019 06:00

Physics, 22.10.2019 06:00

Mathematics, 22.10.2019 06:00




Mathematics, 22.10.2019 06:00

Mathematics, 22.10.2019 06:00

Mathematics, 22.10.2019 06:00

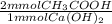 = 15.46 mmol CH₃COOH
= 15.46 mmol CH₃COOH

