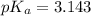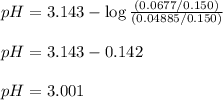Chemistry, 11.06.2021 22:50 robert7248
A buffer solution contains 0.475 M nitrous acid and 0.302 M sodium nitrite . If 0.0224 moles of potassium hydroxide are added to 150 mL of this buffer, what is the pH of the resulting solution

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 01:30
In a spacecraft, the following reaction occurs: co2(g) + 2lioh(s) -> lico3(s) + h2o(i) (i attached picture of equation) how many liters of carbon dioxide will 4 moles of lithium hydroxide (lioh) absorb? (one mole of any gads occupies 22.4 l under certain conditions of temperature and pressure. assume those conditions for this equation.) 45l 6.0l 3.0l 34l
Answers: 1


Chemistry, 22.06.2019 19:30
Helium decays to form lithium. which equation correctly describes this decay?
Answers: 2

Chemistry, 22.06.2019 20:30
A40 kilogram skier starts at the top of a 12 meter high slope. at the bottom, she is travelling 10 meters per second. how much energy does she lose to friction
Answers: 2
You know the right answer?
A buffer solution contains 0.475 M nitrous acid and 0.302 M sodium nitrite . If 0.0224 moles of pota...
Questions



Social Studies, 04.02.2021 20:40

Mathematics, 04.02.2021 20:40




History, 04.02.2021 20:40

Mathematics, 04.02.2021 20:40


Mathematics, 04.02.2021 20:40



Mathematics, 04.02.2021 20:40



Mathematics, 04.02.2021 20:40

Biology, 04.02.2021 20:40


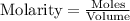 ......(1)
......(1)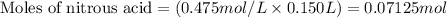
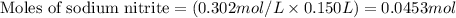
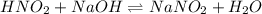
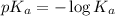 ......(2)
......(2)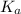 for nitrous acid =
for nitrous acid = 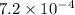
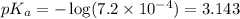
![pH=pK_a+ \log \frac{\text{[conjugate base]}}{\text{[acid]}}](/tpl/images/1372/0490/096b8.png) .......(3)
.......(3)![[NaNO_2]=\frac{0.0677}{0.150}](/tpl/images/1372/0490/ed413.png)
![[HNO_2]=\frac{0.04885}{0.150}](/tpl/images/1372/0490/86d5f.png)