Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2

Chemistry, 22.06.2019 11:10
Which of the following shapes would represent a molecule with two bonded atoms and 3 lone pairs on only one of them , trigonal planar , bent , trigonal pyramidal , linear
Answers: 1

Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3

Chemistry, 22.06.2019 13:50
How does the motion of particles in a gas change as the gas cools
Answers: 2
You know the right answer?
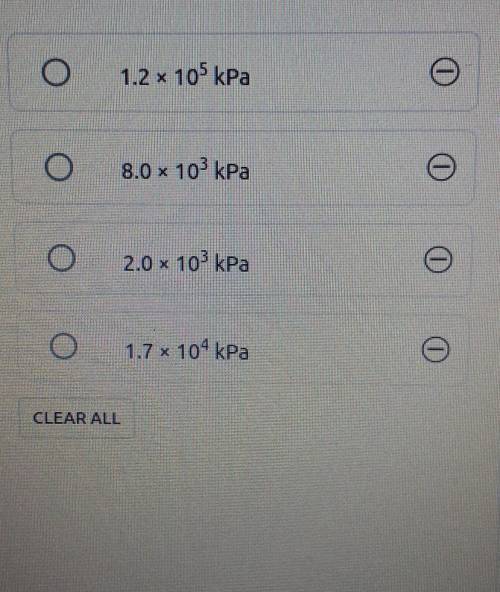
7. A sample of methane gas (CH.) with a mass of 58 g is kept in a 1500 mL container at a temperature...
Questions


Mathematics, 06.11.2020 08:00

Mathematics, 06.11.2020 08:00

Physics, 06.11.2020 08:00



Mathematics, 06.11.2020 08:00

English, 06.11.2020 08:00


Mathematics, 06.11.2020 08:00

Mathematics, 06.11.2020 08:00

Mathematics, 06.11.2020 08:00

English, 06.11.2020 08:00

Mathematics, 06.11.2020 08:00


Mathematics, 06.11.2020 08:00


Chemistry, 06.11.2020 08:00

Mathematics, 06.11.2020 08:00

Mathematics, 06.11.2020 08:00