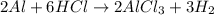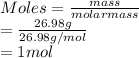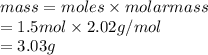Chemistry, 10.06.2021 15:20 drandbone92
Calculate the mass of hydrogen formed when 26.98 g of aluminum reacts with excess hydrochloric acid according to the following balanced chemical equation: 2 Al + 6 HCl → 2 AlCl3 + 3 H2

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:00
If there is any 12 to 14 girls that need a boyfriend just follow me and let me know
Answers: 1

Chemistry, 22.06.2019 09:00
Particles vibrate in a rigid structure and do not move relative to their neighbors.
Answers: 1

Chemistry, 22.06.2019 16:50
What is conserved in the reaction shown below? h2(g) + cl2 (g) --> 2hcl(g)a. mass onlyb. mass and moles onlyc. mass, moles, and molecules onlyd. mass, moles, molecules, and volume
Answers: 2

Chemistry, 22.06.2019 20:40
What effect would average population growth have on land usage? a. urban use of land would rise to more than 30 percent of available land. b. industrial use of land would rise to more than 30 percent of available land. c. the percentage of available land used as cropland would stay the same. d. cropland would fall to about 10 percent of available land.
Answers: 1
You know the right answer?
Calculate the mass of hydrogen formed when 26.98 g of aluminum reacts with excess hydrochloric acid...
Questions





Biology, 27.10.2020 01:00



Social Studies, 27.10.2020 01:00


Mathematics, 27.10.2020 01:00

Mathematics, 27.10.2020 01:00

Mathematics, 27.10.2020 01:00


Biology, 27.10.2020 01:00


Mathematics, 27.10.2020 01:00




Mathematics, 27.10.2020 01:00