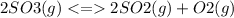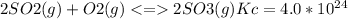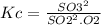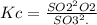Chemistry, 09.06.2021 14:00 slawson4328
The value for the equilibrium constant for the reaction 2SO2(g)+O2(g) rightleftharpoons 2SO3(g) is 4.0*10^ 1 24 at 298K What would be the value for the equilibrium constant for the following reaction at the same temperature? 2SO3(g) rightleftharpoons2502(g)+O2(g)

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:30
Start an single atom tab. observe the decay of polonium-211. after each decay, press the reset nucleus button to watch the process again. write a description of alpha decay for po-211
Answers: 2

Chemistry, 22.06.2019 10:00
In a water molecule, hydrogen and oxygen are held together by a(an) bond. a) double covalent b) ionic c) nonpolar covalent d) hydrogen e) polar covalent
Answers: 1

Chemistry, 22.06.2019 10:00
Drug abuse will not lead to physical and psychological dependence. true or false ?
Answers: 2

Chemistry, 22.06.2019 10:10
How do you identify the anode on a power source such as a battery? how do you identify the cathode? how are terms anion and cation?
Answers: 1
You know the right answer?
The value for the equilibrium constant for the reaction 2SO2(g)+O2(g) rightleftharpoons 2SO3(g) is 4...
Questions

Health, 04.08.2019 21:20


History, 04.08.2019 21:20

Social Studies, 04.08.2019 21:20

Mathematics, 04.08.2019 21:20



Biology, 04.08.2019 21:20


Social Studies, 04.08.2019 21:20

English, 04.08.2019 21:20


History, 04.08.2019 21:20

Mathematics, 04.08.2019 21:20

Social Studies, 04.08.2019 21:20

Spanish, 04.08.2019 21:20

Advanced Placement (AP), 04.08.2019 21:20



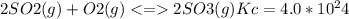 at 298K .
at 298K .