
Chemistry, 09.06.2021 09:50 aliceohern
A solution containing 1.22 g of a diprotic acid H2CH2O4 (malonic acid)
was titrated with 45.5 mL of NaOH to reach the second equivalence
point. What is the concentration of the NaOH solution? (MW_malonic
acid = 104.06 g/mol)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:00
Which of these would be caused by a chemical change? a) the formation of lava. b) sedimantary rock layering over time. c) metamorphic rock forming from igneous. d) metamorphic rock eroding to form sedimentary rock.
Answers: 3

Chemistry, 22.06.2019 20:00
The picture represents the process that produces most of the energy used by living organisms on earth. which process is represented in the picture? a) the magnetic attraction between two hydrogen nuclei. b) the fusion of hydrogen nuclei to produce a helium nucleus in the core of the sun. c) the fission of hydrogen nuclei to produce a helium nucleus in the core of the sun. d) the chemical reaction between hydrogen nuclei to produce a helium nucleus in earth's atmosphere.
Answers: 3

Chemistry, 22.06.2019 23:10
Match the formula for the following compound: magnesium sulfate heptahydratemgs·7h2omg2so4·7h2omg(so4)2·7h2omgso4·7h2o
Answers: 1

Chemistry, 23.06.2019 03:10
Which of the following compounds would be expected to have the strongest ionic bonds? a)the compound that has b)the largest ions with the greatest charge c)the compound that has d)the largest ions with the least charge the compound that has the smallest ions with the greatest charge the compound that has the smallest ions with the least charge
Answers: 2
You know the right answer?
A solution containing 1.22 g of a diprotic acid H2CH2O4 (malonic acid)
was titrated with 45.5 mL of...
Questions


Mathematics, 13.07.2019 12:30

Mathematics, 13.07.2019 12:30

English, 13.07.2019 12:30








English, 13.07.2019 12:30

Mathematics, 13.07.2019 12:30

Mathematics, 13.07.2019 12:30


Mathematics, 13.07.2019 12:30


Mathematics, 13.07.2019 12:30


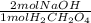 = 0.02344 mol NaOH
= 0.02344 mol NaOH

