
Chemistry, 09.06.2021 03:00 blkrosesss
Calculate the number of milliliters of 0.587 M NaOH required to precipitate all of the Fe3 ions in 197 mL of 0.654 M FeCl3 solution as Fe(OH)3. The equation for the reaction is: FeCl3(aq) 3NaOH(aq) Fe(OH)3(s) 3NaCl(aq)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Gymnast always perform on padded mats. how does the mats protect the gymnast
Answers: 2

Chemistry, 22.06.2019 04:50
The name of the ion, s2-, is: sulfurous ion sulfide ion sulfur ion sulfate ion
Answers: 1


Chemistry, 22.06.2019 09:00
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1
You know the right answer?
Calculate the number of milliliters of 0.587 M NaOH required to precipitate all of the Fe3 ions in 1...
Questions


Mathematics, 23.01.2021 07:20

Spanish, 23.01.2021 07:20

Mathematics, 23.01.2021 07:20

Chemistry, 23.01.2021 07:20

English, 23.01.2021 07:20

Mathematics, 23.01.2021 07:20



Health, 23.01.2021 07:20


Social Studies, 23.01.2021 07:20

Mathematics, 23.01.2021 07:20

Social Studies, 23.01.2021 07:20



Mathematics, 23.01.2021 07:30

Health, 23.01.2021 07:30


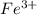 ions in 197 mL of 0.654 M
ions in 197 mL of 0.654 M 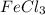 solution as
solution as 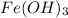 .
.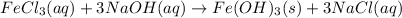
 Volume (in L)
Volume (in L)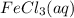 reacts with 3 moles of NaOH(aq). Hence, moles of
reacts with 3 moles of NaOH(aq). Hence, moles of 

