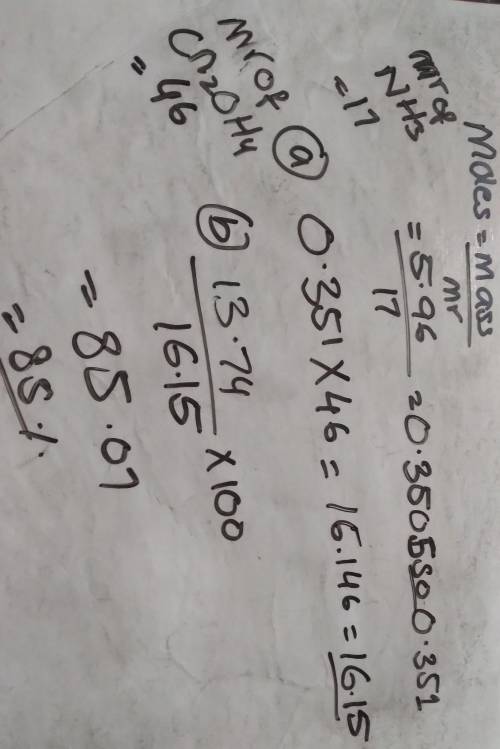Chemistry, 05.06.2021 23:00 deaishaajennings123
5.96 g of ammonia reacts completely according to the following reaction:
2 NH3, (g) + Co2, (g) → CN2,OH4, (s) + H20 (l)
(a) What is the theoretical yield of urea (CN, OH,) for this reaction?
(b) If 13.74 g of urea are produced, what is the percent yield for this equation?
please show work, will give brainliest

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:30
Summarize possible ways in which phases of matter could combine to form a solution.
Answers: 2

Chemistry, 22.06.2019 08:30
How would the number of moles (n) of o2 change if the atmospheric pressure doubled but all other variables stayed the same
Answers: 2

Chemistry, 22.06.2019 22:00
What mass of glucose is produced when 54g of water react with carbon dioxide
Answers: 1

Chemistry, 23.06.2019 06:00
What are the coefficients to balance the following equation? ba+br2=babr2
Answers: 2
You know the right answer?
5.96 g of ammonia reacts completely according to the following reaction:
2 NH3, (g) + Co2, (g) → CN...
Questions

Mathematics, 20.05.2020 20:58

Chemistry, 20.05.2020 20:58


Mathematics, 20.05.2020 20:59

Mathematics, 20.05.2020 20:59

Chemistry, 20.05.2020 20:59

Social Studies, 20.05.2020 20:59



History, 20.05.2020 20:59

Mathematics, 20.05.2020 20:59

Mathematics, 20.05.2020 20:59

History, 20.05.2020 20:59





SAT, 20.05.2020 20:59

Social Studies, 20.05.2020 20:59

Mathematics, 20.05.2020 20:59