Al3+(g)+3e + Al(s)

Chemistry, 05.06.2021 02:30 donavancoulter712
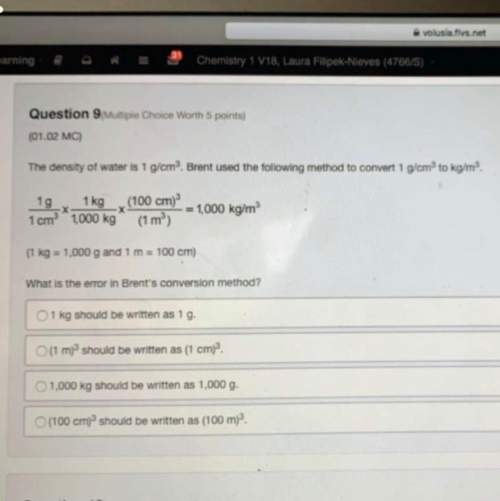
Read the table below.
Reduction Half-Reaction: Mn2+ (aq) + 2e → Mn(s)
Al3+(g)+3e + Al(s)
(v)
-1.18
-1.66
Which reactants would lead to a spontaneous reaction?
O Mn2+ and AI
O Mn and A13+
O Al and Mn
O Mn2+ and A13+

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 15:00
If a planet rotates 360 degrees during a 24 hour time period, what does that tell us about the planet? a. the middle of the planet is in darkness b. the seasons on the planet vary every day. c. the planet runs on a 12-hour time clock. d. the temperature on the planet varies daily.
Answers: 1

Chemistry, 21.06.2019 22:40
How many electrons does silver have to give up in order to achieve a sido noble gas electron configuration
Answers: 1

Chemistry, 22.06.2019 04:30
Use the drop-down menus to answer each question. which runner finished the 100 m race in the least amount of time? which runner stopped running for a few seconds during the race? at what distance did anastasia overtake chloe in the race?
Answers: 1

Chemistry, 22.06.2019 13:00
Using the thermodynamic information in the aleks data tab, calculate the standard reaction free energy of the following chemical reaction: →+p4o10s6h2ol4h3po4s round your answer to zero decimal places.
Answers: 3
You know the right answer?
Read the table below.
Reduction Half-Reaction: Mn2+ (aq) + 2e → Mn(s)
Al3+(g)+3e + Al(s)
Al3+(g)+3e + Al(s)
Questions






Mathematics, 19.10.2019 13:30

English, 19.10.2019 13:30


Mathematics, 19.10.2019 13:30

Biology, 19.10.2019 13:30

Health, 19.10.2019 13:30



English, 19.10.2019 13:30

Business, 19.10.2019 13:30

Mathematics, 19.10.2019 13:30



History, 19.10.2019 13:30

English, 19.10.2019 13:30