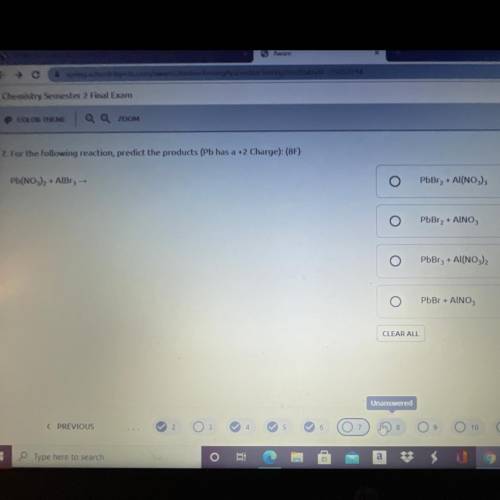
Pb(NO3)2 + AlBr3 →


Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 20:10
Suppose you mix one mole of sulfuric acid (h2so4) with 1 mole of sodium hydroxide(naoh). why does the ph of the solution remain below 7? ( explain so i can get better understanding! )
Answers: 2

Chemistry, 22.06.2019 22:10
Determine the ph of 0.10 m nh3 solution. nh3 is a weak base with a kb equal to 1.8 x 10-5 round answer to nearest whole number.
Answers: 1

Chemistry, 23.06.2019 01:00
Chromium(iii) sulfate is a transition metal compound containing the metal chromium and the polyatomic ion sulfate. the oxidation state of chromium in this compound is , and the chemical formula of the compound is ( ) . reset next
Answers: 3
You know the right answer?
For the following reaction, predict the products (Pb has a +2 Charge):
Pb(NO3)2 + AlBr3 →
Pb(NO3)2 + AlBr3 →
Questions

Social Studies, 13.01.2021 22:10




English, 13.01.2021 22:10


French, 13.01.2021 22:10

Mathematics, 13.01.2021 22:10

Mathematics, 13.01.2021 22:10

Social Studies, 13.01.2021 22:10

History, 13.01.2021 22:10



Chemistry, 13.01.2021 22:10

Mathematics, 13.01.2021 22:10


Mathematics, 13.01.2021 22:10



Mathematics, 13.01.2021 22:10