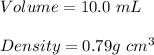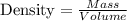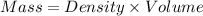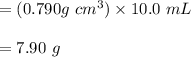A chemistry student needs of acetone for an experiment. By consulting the CRC Handbook of Chemistry and Physics, the student discovers that the density of acetone is . Calculate the volume of acetone the student should pour out. Be sure your answer has the correct number of significant digits.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 11:50
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3

Chemistry, 22.06.2019 21:00
Which answer tells the reason the earth’s climate is getting warmer? too many animals are becoming extinct. large glaciers are melting in antarctica. the earth is moving closer to the sun. driving cars gives off gases that trap heat in the atmosphere.
Answers: 1

Chemistry, 22.06.2019 23:50
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3
You know the right answer?
A chemistry student needs of acetone for an experiment. By consulting the CRC Handbook of Chemistry...
Questions

Biology, 08.07.2020 14:01

Mathematics, 08.07.2020 14:01


Biology, 08.07.2020 14:01

Mathematics, 08.07.2020 14:01

Mathematics, 08.07.2020 14:01

Mathematics, 08.07.2020 14:01

Mathematics, 08.07.2020 14:01


Mathematics, 08.07.2020 14:01

Mathematics, 08.07.2020 14:01



Social Studies, 08.07.2020 14:01


Mathematics, 08.07.2020 14:01




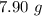 ".
".