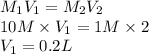A 10 M concentrated stock solution of NaCl is used to prepare 2 liters of diluted 1 M
solution. Which of the following is true for the process used to achieve the required
dilution? (5 points)
O The volume of the solvent used is less than 0.1 liters.
O The volume of stock solution used is less than 0.1 liters.
The volume of stock solution used is more than 2 liters.
The volume of the solvent used is less than 2 liters.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 12:00
In a laboratory, 1.55mg of an organic compound containing carbon, hydrogen, and oxygen is burned for analysis. this combustion resulted in the formation of 1.45mg of carbon dioxide and .89 mg of water. what is the empirical formula for this compound?
Answers: 1

Chemistry, 22.06.2019 20:30
Which of the following is not true about the atomic model of substances?
Answers: 1

Chemistry, 22.06.2019 21:20
One way in which the useful metal copper is produced is by dissolving the mineral azurite, which contains copper(ii) carbonate, in concentrated sulfuric acid. the sulfuric acid reacts with the copper(ii) carbonate to produce a blue solution of copper(ii) sulfate. scrap iron is then added to this solution, and pure copper metal precipitates out because of the following chemical reaction: (s) (aq) (s) (aq) suppose an industrial quality-control chemist analyzes a sample from a copper processing plant in the following way. he adds powdered iron to a copper(ii) sulfate sample from the plant until no more copper will precipitate. he then washes, dries, and weighs the precipitate, and finds that it has a mass of .
Answers: 2
You know the right answer?
A 10 M concentrated stock solution of NaCl is used to prepare 2 liters of diluted 1 M
solution. Whi...
Questions




History, 25.11.2019 08:31

Biology, 25.11.2019 08:31

Social Studies, 25.11.2019 08:31

Mathematics, 25.11.2019 08:31

Mathematics, 25.11.2019 08:31

History, 25.11.2019 08:31

Arts, 25.11.2019 08:31

Social Studies, 25.11.2019 08:31


English, 25.11.2019 08:31


Biology, 25.11.2019 08:31



English, 25.11.2019 08:31

Mathematics, 25.11.2019 08:31

Geography, 25.11.2019 08:31

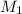 = 10 M,
= 10 M, 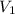 = ?
= ?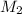 = 1 M,
= 1 M, 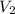 = 2 L
= 2 L