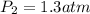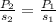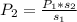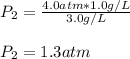Chemistry, 31.05.2021 05:20 Carrchris021
If the solubility of a gas in water is 3.0 g/L when the pressure of the gas above the water is 4.0 atm, what is the pressure of the gas above the water when the solubility of the gas is 1.0 g/L

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1


Chemistry, 22.06.2019 12:00
Explain what happens at the saturation point when adding salt to water at room temperature.
Answers: 1

You know the right answer?
If the solubility of a gas in water is 3.0 g/L when the pressure of the gas above the water is 4.0 a...
Questions

Geography, 11.12.2020 01:00




History, 11.12.2020 01:00

History, 11.12.2020 01:00


Mathematics, 11.12.2020 01:00

Arts, 11.12.2020 01:00



Mathematics, 11.12.2020 01:00

Mathematics, 11.12.2020 01:00

Mathematics, 11.12.2020 01:00




Mathematics, 11.12.2020 01:00

Mathematics, 11.12.2020 01:00