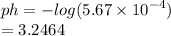b. 3.25

Chemistry, 30.05.2021 23:00 relliott4950
What is the pH of a solution with an H3O+ concentration of 5.67 x 10-4 M?
a. 1.00
b. 3.25
C. -1.00
d. -3.25

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1

Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 14:00
650.j is the same amount of energy as? 2720cal1550cal650.cal2.72cal
Answers: 2

Chemistry, 22.06.2019 19:30
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2
You know the right answer?
What is the pH of a solution with an H3O+ concentration of 5.67 x 10-4 M?
a. 1.00
b. 3.25
b. 3.25
Questions

Mathematics, 17.07.2019 03:00

History, 17.07.2019 03:00


History, 17.07.2019 03:00

History, 17.07.2019 03:00

Social Studies, 17.07.2019 03:00





Mathematics, 17.07.2019 03:00

Computers and Technology, 17.07.2019 03:00

History, 17.07.2019 03:00


Mathematics, 17.07.2019 03:00

Mathematics, 17.07.2019 03:00



Mathematics, 17.07.2019 03:00

Mathematics, 17.07.2019 03:00

![pH = - log [ { H_3O}^{+}]](/tpl/images/1355/8555/1a2ed.png)