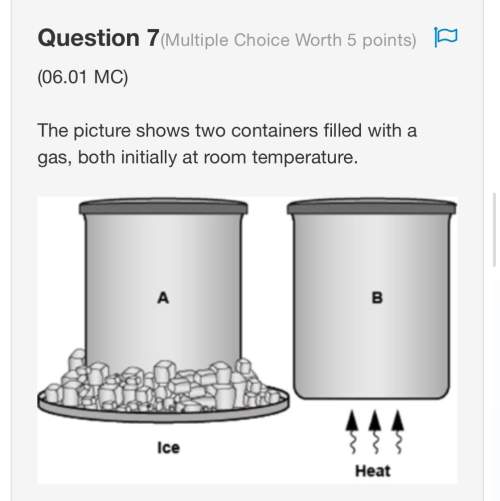
Chemistry, 30.05.2021 04:00 adjjones2011
A standard lanthanum solution is prepared by dissolving 0.1968 grams of lanthanum oxide (La2O3) in excess nitric acid and diluting to one liter in a volumetric flask. Calculate the concentration of the solution expressed as molarity of lanthanum nitrate and as ppm lanthanum.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:20
Complete the table for ion charge based upon their losing or gaining electrons in the outer shell. (use the periodic table as necessary.) group most likely ionic charge # of valence electrons i +1 ii +2 iii +3 iv +4 or -4 v -3 vi -2 vii -1 viii 0
Answers: 2

Chemistry, 22.06.2019 15:00
Which of the following is the correct formula for copper (i) sulfate trihydrate? cuso4 · 3h2o cuso4(h2o)3 cu2so4(h2o)3 cu2so4 · 3h2o
Answers: 1

Chemistry, 23.06.2019 01:20
How can parts of a solution be separated by chromatography?
Answers: 1

Chemistry, 23.06.2019 02:00
Now look at the segment of the graph between the two data points marked with black squares. describe how the boiling point and melting point plots behave between these points. be as specific as possible.
Answers: 1
You know the right answer?
A standard lanthanum solution is prepared by dissolving 0.1968 grams of lanthanum oxide (La2O3) in e...
Questions

Mathematics, 02.03.2021 22:00



English, 02.03.2021 22:00

Mathematics, 02.03.2021 22:00

Geography, 02.03.2021 22:00


Mathematics, 02.03.2021 22:00

English, 02.03.2021 22:00

Mathematics, 02.03.2021 22:00

English, 02.03.2021 22:00


English, 02.03.2021 22:00



History, 02.03.2021 22:00

Mathematics, 02.03.2021 22:00

Biology, 02.03.2021 22:00


Mathematics, 02.03.2021 22:00