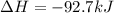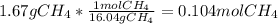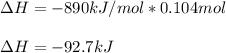Chemistry, 29.05.2021 17:00 allenpaietonp9v8sv
When 1 mol of methane is burned at constant pressure, −890 kJ/mol of energy is released as heat. If a 1.67 g sample of methane is burned at constant pressure, what will be the value of ∆H

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:00
Americium-241 undergoes fission to produce three neutrons per fission event. if a neutron-absorbing material is mixed in with this sample so that the rate of neutron production drops down to 1.8 neutrons per fission event, which will be effective at achieving a critical mass? check all that apply. remove a deflective shield surrounding the sample. remove absorbent material mixed in with the sample. compress the sample of americium-241.
Answers: 1

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1

Chemistry, 22.06.2019 22:00
Scientists often have to deal with numbers that are either very large or very small. for example, the radius of the sun is approximately 696,000 kilometers, while bacterial cells are as small as 1.9 × 10-4 millimeters. express each number in an alternate form.
Answers: 1

Chemistry, 23.06.2019 07:30
Which of the following statements best explains why chemistry is testable a) it can measure data by experiments b) it cannot add new evidence c) it cannot be verified d) it is biased
Answers: 1
You know the right answer?
When 1 mol of methane is burned at constant pressure, −890 kJ/mol of energy is released as heat. If...
Questions



Mathematics, 14.07.2019 00:30



Mathematics, 14.07.2019 00:30



Mathematics, 14.07.2019 00:30

Mathematics, 14.07.2019 00:30

Mathematics, 14.07.2019 00:30



Mathematics, 14.07.2019 00:30

Mathematics, 14.07.2019 00:30



Mathematics, 14.07.2019 00:30