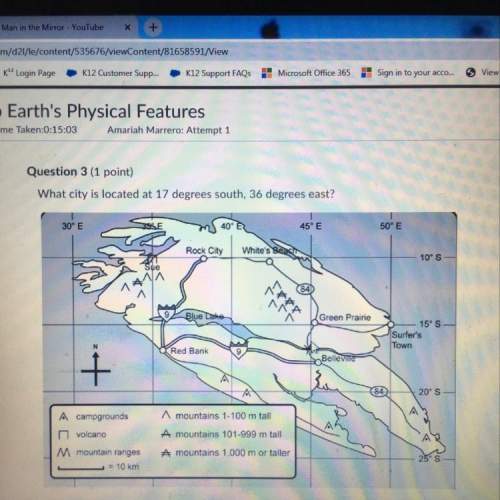
Chemistry, 28.05.2021 14:00 abdulbasharee99
State the change in oxidation number of sulphur in the redox reaction below: I2+2S2O3² 2I +S4O7²

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 15:50
Where are chemicals found at work? a. only in cleaning products b. only in carpets and paint c. in every area of work d. only in food preparation submit
Answers: 1

Chemistry, 22.06.2019 04:00
Write the balanced equation for a reaction between aqueous nitric acid (hno3) and solid lithium metal (this is a single replacement reaction)
Answers: 1

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 molarity koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 11:00
When hydrochloric acid reacts with potassium hydroxide solution, the following reaction occurs. hcl (aq) + koh (aq) h2o (l) + kcl (aq) the reaction gives off heat energy, so it is an reaction.
Answers: 1
You know the right answer?
State the change in oxidation number of sulphur in the redox reaction below: I2+2S2O3²
2I +S4O7²...
Questions







History, 30.06.2019 18:50

Business, 30.06.2019 18:50


History, 30.06.2019 18:50


History, 30.06.2019 18:50


Spanish, 30.06.2019 18:50

Social Studies, 30.06.2019 18:50


History, 30.06.2019 19:00


English, 30.06.2019 19:00

Mathematics, 30.06.2019 19:00