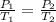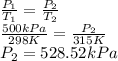Chemistry, 28.05.2021 03:50 ttangelique
A car starts out when the air temperature is 298 K and the absolute (total) air pressure in the tires is 500 kPa. After driving a while, the temperature of the air in the tires increases to 315 K. What is the pressure in the tires at that point, assuming their volume does not change

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:30
Un cierto gas tiene un volumen de 800ml a 80°c y 600ml a 80°c y 600mmhg de presión. ¿cual será el volumen del gas a condiciones normales? sí el gas es oxígeno, ¿cuál será su peso? y ¿cuántas moléculas están presentes en el sistema?
Answers: 2

Chemistry, 22.06.2019 08:00
What are the similarities of physical and chemical change ?
Answers: 1


Chemistry, 22.06.2019 16:10
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1
You know the right answer?
A car starts out when the air temperature is 298 K and the absolute (total) air pressure in the tire...
Questions


History, 20.01.2021 22:40


Mathematics, 20.01.2021 22:40


Mathematics, 20.01.2021 22:40

Law, 20.01.2021 22:40



English, 20.01.2021 22:40



Social Studies, 20.01.2021 22:40


Mathematics, 20.01.2021 22:40

Chemistry, 20.01.2021 22:40

Mathematics, 20.01.2021 22:40

Arts, 20.01.2021 22:50


Mathematics, 20.01.2021 22:50

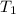 = 500 K,
= 500 K, 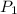 = 500 kPa
= 500 kPa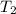 = 315 K,
= 315 K, 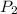 = ?
= ?