Chemistry, 27.05.2021 22:50 rachellynn02
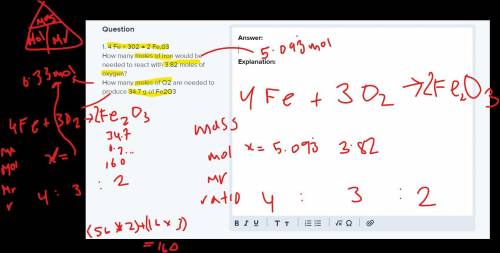
1. 4 Fe + 302 → 2 Fe,03
How many moles of iron would be needed to react with 3.82 moles of oxygen?
How many moles of O2 are needed to produce 34.7 g of Fe2O3

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 14:10
16. in a reaction that has reached equilibrium, a. the forward and reverse reactions are occurring at the same rate. b. the reactants and products are in equal concentrations. c. the forward reaction has gone further than the reverse reaction. d. there are equal numbers of atoms on both sides of the equation. e. a, b, and d are correct.
Answers: 2

Chemistry, 22.06.2019 20:00
For the reaction c6h14(g) & longrightarrow; c6h6(g) + 4h2(g), δp(h2)/δt was found to be 2.5 x 10-2 atm/s, where δp(h2) is the change in pressure of hydrogen. determine δp(c6h14)/δt for this reaction at the same time.
Answers: 2

You know the right answer?
1. 4 Fe + 302 → 2 Fe,03
How many moles of iron would be needed to react with 3.82 moles of oxygen?<...
Questions


Mathematics, 09.12.2020 17:30

Physics, 09.12.2020 17:30






Business, 09.12.2020 17:30



Chemistry, 09.12.2020 17:30


Physics, 09.12.2020 17:30


Mathematics, 09.12.2020 17:30




Mathematics, 09.12.2020 17:30