Chemistry, 27.05.2021 22:40 microwave13016
1. For each of the following formulas:
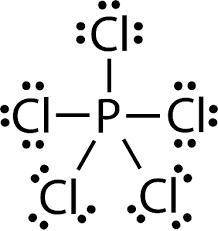
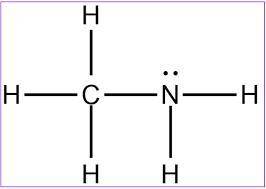
1) if ionic, write the formulas of the ions; if covalent, draw the Lewis structure
2) For each covalent compound, describe the electronic and molecular geometry
3) For each covalent compound, describe the hybridization of the central atom
4) Name each compound, except the organic one.
5) How many sigma and how many pi bonds does each compound have?
MnSO4 CH3NH2 PCl5 O2 LiF


Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:10
How many moles of gas are present if p=11 atm, v=12l, t=185k?
Answers: 1

Chemistry, 22.06.2019 07:30
What three things determine the shape and size of a puddle when water is poured out onto a surface
Answers: 2


Chemistry, 23.06.2019 07:00
Explain what happened when the storm surges from hurricanes reached the gulf coast
Answers: 1
You know the right answer?
1. For each of the following formulas:
1) if ionic, write the formulas of the ions; if covalent, dr...
Questions

Mathematics, 22.03.2020 21:08


Arts, 22.03.2020 21:08

Mathematics, 22.03.2020 21:09

Mathematics, 22.03.2020 21:09

Mathematics, 22.03.2020 21:09





History, 22.03.2020 21:09




History, 22.03.2020 21:09