
Chemistry, 27.05.2021 21:40 sammizwang
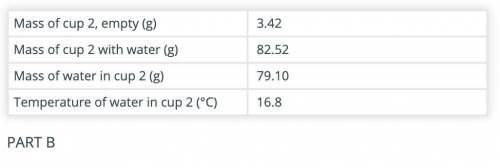
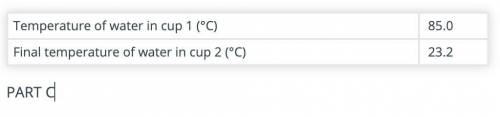
Calculate the amount of heat gained by the water in cup 2 after adding the hot object(s) to it.
Use the data recorded in parts B and C and the formula Q = mCΔT. The specific heat capacity of water is 4.186 J/(g °C).
(part B and C numbers are included)

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Ahypothrticalax type of ceramic material is known to have a density of 2.10 g/cm3 and a unit cell of cubic symmetry with a cell edge length of 0.57 nm. the atomic weights of the a and x elements are 28.5and 30.0 g/mol, respectively. on the basis of this information, which of the following crystal structures is (are) possible for this material: sodium chloride, cesium chloride, or zinc blende
Answers: 1



Chemistry, 23.06.2019 05:00
What is dhmo? hint: you find it everywhere something is wet..
Answers: 1
You know the right answer?
Calculate the amount of heat gained by the water in cup 2 after adding the hot object(s) to it.
Use...
Questions






Mathematics, 04.09.2020 19:01












Mathematics, 04.09.2020 19:01




