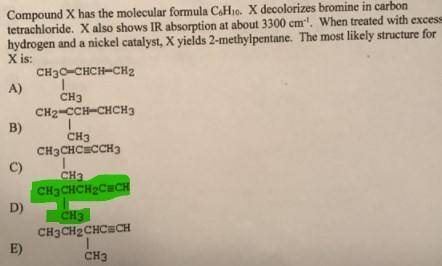Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Which type of bonding involves the complete transfer of a valence electron from a less electrogrative atom to a more electronegative one
Answers: 1


Chemistry, 23.06.2019 04:40
6) (a) calculate the absorbance of the solution if its concentration is 0.0278 m and its molar extinction coefficient is 35.9 l/(mol cm). the depth of the cell is 5 mm. (b) what is the %t? (7) calculate the absorbance of the solution if the transmitted light intensity is 70% of the initial light beam intensity
Answers: 1

Chemistry, 23.06.2019 05:50
What are the coefficients to balance the following equation? ba+br=babr2
Answers: 1
You know the right answer?
Compound X has the molecular formula C6H10. X decolorizes bromine in carbon tetrachloride. X also sh...
Questions

Mathematics, 13.04.2021 01:30


Geography, 13.04.2021 01:30




Mathematics, 13.04.2021 01:30

Medicine, 13.04.2021 01:30

English, 13.04.2021 01:30




Mathematics, 13.04.2021 01:30

Mathematics, 13.04.2021 01:30




Chemistry, 13.04.2021 01:30


Mathematics, 13.04.2021 01:40