
Chemistry, 26.05.2021 01:00 emilystartk
) Dinitrogen Tetroxide partially decomposes according to the following equilibrium: N2O4 (g) 2NO2 (g) A 1.00-L flask is charged with 0.400 mol of N2O4. At equilibrium at 373 K, 0.0055 mol of N2O4 remains. Keq for this reaction is .

Answers: 3
Another question on Chemistry


Chemistry, 21.06.2019 20:30
Five students had to answer the question how are elements arranged in a periodic tabledamon: i think the elements are arranged by increasing massflo: i think the elements are arranged according to their properties sienna: i think the elements are arranged by when their discovers kyle: i think the elements are arranged according to how common they areglenda: i don't agree with any of themwho is right
Answers: 1

Chemistry, 21.06.2019 21:00
Consider the nuclear equation below. 239 > x + 4 he 94 2 what is x? 1.235 cm 96 2.243 u 92 3.235 u 92 4.243 cm 96
Answers: 2

Chemistry, 22.06.2019 14:30
Amixture that has two or more substances that are spread out evenly is called a. compound b. heterogeneous c. substance d. homogeneous
Answers: 1
You know the right answer?
) Dinitrogen Tetroxide partially decomposes according to the following equilibrium: N2O4 (g) 2NO2 (g...
Questions

Mathematics, 31.03.2020 23:28

Physics, 31.03.2020 23:28

Mathematics, 31.03.2020 23:28

Mathematics, 31.03.2020 23:28

Biology, 31.03.2020 23:28


English, 31.03.2020 23:28

Physics, 31.03.2020 23:28

Computers and Technology, 31.03.2020 23:28

Mathematics, 31.03.2020 23:28


Chemistry, 31.03.2020 23:28


Mathematics, 31.03.2020 23:28

History, 31.03.2020 23:28

Mathematics, 31.03.2020 23:28

Mathematics, 31.03.2020 23:28

Biology, 31.03.2020 23:28


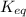 for this reaction is 1.578.
for this reaction is 1.578.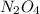 = 0.4 mol
= 0.4 mol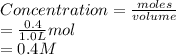
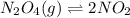
![[NO_{2}]](/tpl/images/1347/8951/53e25.png) is calculated as follows.
is calculated as follows.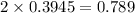
![K_{eq} = \frac{[NO_{2}]^{2}}{[N_{2}O_{4}]}\\= \frac{(0.789)^{2}}{(0.3945)}\\= 1.578](/tpl/images/1347/8951/12c3a.png)


