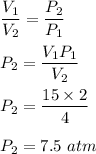Chemistry, 25.05.2021 22:30 briannagotfanz
A sample of krypton occupies 15.0 L at a pressure of 2.1 atm. Use Boyle's Law to find the pressure of the krypton when the volume is decreased to 4L.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 19:50
Which sentence from holes contains an implied personality trait? stanley and his parents had tried to pretend that he was just going away to camp for a while, just like rich kids do. he'd just been in the wrong place at the wrong time. stanley felt somewhat dazed as the guard unlocked his handcuffs and led him off the bus. stanley nodded to show he understood
Answers: 3

Chemistry, 23.06.2019 00:10
Find the missing probability in the table below a.0.10 b.40 c.0.80 d. 0.20
Answers: 2


Chemistry, 23.06.2019 10:30
Ireally need ! calcium metal reacts with a potassium chloride solution to form calcium chloride and potassium ions. balance this reaction. (s) + (aq) → cacl2(s) + +(aq) a) 1, 2, 1, 2 b) 1, 2, 1, 1 c) 1, 1, 1, 1 d) 2, 1, 2, 1
Answers: 1
You know the right answer?
A sample of krypton occupies 15.0 L at a pressure of 2.1 atm. Use Boyle's Law to find the pressure o...
Questions

History, 26.05.2021 19:00

Chemistry, 26.05.2021 19:00


Mathematics, 26.05.2021 19:00

Mathematics, 26.05.2021 19:00

World Languages, 26.05.2021 19:00

History, 26.05.2021 19:00



Business, 26.05.2021 19:00





English, 26.05.2021 19:00

English, 26.05.2021 19:00


Mathematics, 26.05.2021 19:00


Mathematics, 26.05.2021 19:00