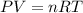Chemistry, 25.05.2021 19:00 beanokelley
How many moles of gas would you have if you have a volume of 38.0 L under a pressure of 1430 mmHg at standard temperature

Answers: 1
Another question on Chemistry



Chemistry, 22.06.2019 22:30
Amedication is given at a dosage of 3.000 mg of medication per kg of body weight. if 0.1500 g of medication is given, then what was the patient's weight in pounds (lbs)? there are 453.59g in 1 lb.
Answers: 2

Chemistry, 22.06.2019 22:30
What relationship exists between an enzyme and a catalyst?
Answers: 1
You know the right answer?
How many moles of gas would you have if you have a volume of 38.0 L under a pressure of 1430 mmHg at...
Questions


English, 09.11.2020 21:40




Mathematics, 09.11.2020 21:40

History, 09.11.2020 21:40


Chemistry, 09.11.2020 21:40


Mathematics, 09.11.2020 21:40


Chemistry, 09.11.2020 21:40

History, 09.11.2020 21:40

Mathematics, 09.11.2020 21:40



Mathematics, 09.11.2020 21:40

English, 09.11.2020 21:40

Mathematics, 09.11.2020 21:40