
Chemistry, 25.05.2021 01:00 NetherisIsTheQueen
In an aqueous chloride solution cobalt(II) exists in equilibrium with the complex ion CoCl42-. Co2 (aq) is pink and CoCl42-(aq) is blue. At Low Temperature the pink color pre-dominates. At High Temperature the blue color is strong. If we represent the equilibrium as:
CoCl4^2-(aq) <--> Co2+(aq) + 4Cl-(aq)
We can conclude that:___.
1. This reaction is:___.
A. Exothermic
B. Endothermic
C. Neutral
2. When the temperature is decreased the equilibrium constant, K:.
A. Increases
B. Decreases
C. Remains the same.
3. When the temperature is decreased the equilibrium concentration of Co2:.
A. Increases
B. Decreases
C. Remains the same.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 18:00
The fact that the total amount of energy in a system remains constant is a(n)
Answers: 1

Chemistry, 22.06.2019 22:00
11) burning your hand when accidentally touching a hot plate is an example of which heat transfer? a. conduction b. convection c. radiation d. none of these
Answers: 2

Chemistry, 22.06.2019 23:00
What does a numerical subscript following an element in a chemical formula mean?
Answers: 1
You know the right answer?
In an aqueous chloride solution cobalt(II) exists in equilibrium with the complex ion CoCl42-. Co2 (...
Questions




Mathematics, 02.08.2021 18:00


English, 02.08.2021 18:00

Mathematics, 02.08.2021 18:00


English, 02.08.2021 18:00


Mathematics, 02.08.2021 18:00





Physics, 02.08.2021 18:00


Chemistry, 02.08.2021 18:00


Mathematics, 02.08.2021 18:00

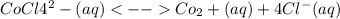
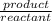 =
=![\frac{[CO^2^+][Cl^-^4]}{CoCl^2^-_4}](/tpl/images/1345/2628/18cbc.png)
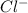 and
and  rises, and K rises as well , thus it increases .
rises, and K rises as well , thus it increases .

