
Chemistry, 24.05.2021 23:30 rivermadds4163
Help plz:)))I’ll mark u Brainliest Part 3 Directions : Identify the type of chemical reaction from the UNbalanced equations : (synthesis single replacement , double replacement, decomposition combustion.)
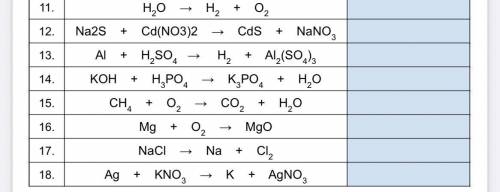

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 21:50
Given the data below for the reaction, 2 a + 2 b + 4 c => d + e + 3 f, the reaction is fill in the [ ] order in a, fill in the [ ] order in b, fill in the [ ] order in c and fill in the [ ] order overall. (use the words "first, second, third, fourth" to fill each blank)experimentinitial conc of a, mol/l initial conc of b, mol/l initial conc of c, mol/l initial rate, mol/l.s1 0.1 0.1 0.2 2 x 10-32 0.2 0.3 0.2 6 x 10-33 0.3 0.1 0.2 2 x 10-34 0.4 0.3 0.4 1.2 x 10-2
Answers: 2


Chemistry, 23.06.2019 00:30
The molecular weight of carbon dioxide, co2, is 44.00 amu, and the molecular weight of nitrous dioxide, no2, is 46.01 amu, so no2 diffuses co2
Answers: 2
You know the right answer?
Help plz:)))I’ll mark u Brainliest
Part 3 Directions : Identify the type of chemical reaction from...
Questions

English, 10.11.2020 07:40


Health, 10.11.2020 07:40

Mathematics, 10.11.2020 07:40

English, 10.11.2020 07:40

History, 10.11.2020 07:40

History, 10.11.2020 07:40

Health, 10.11.2020 07:40

History, 10.11.2020 07:40

Mathematics, 10.11.2020 07:40



Mathematics, 10.11.2020 07:40





Mathematics, 10.11.2020 07:40

Mathematics, 10.11.2020 07:40

Social Studies, 10.11.2020 07:40



