
Chemistry, 24.05.2021 18:20 lizbethmillanvazquez
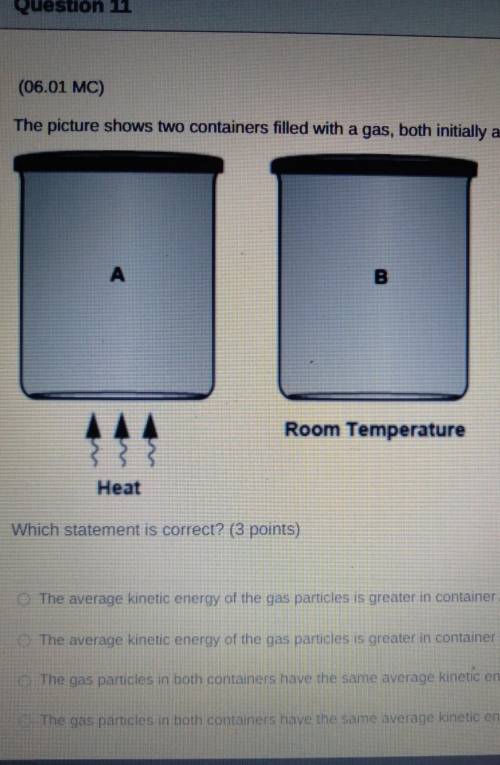
The picture shows two containers filled with a gas, both initially at room temperature.
A Heat
B Room Temperature
Which statement is correct?
The average kinetic energy of the gas particles is greater in container A because its particles move faster.
The average kinetic energy of the gas particles is greater in container B because it has a lower temperature.
The gas particles in both containers have the same average kinetic energy because they have the same volume.
The gas particles in both containers have the same average kinetic energy because they have equal number of particles

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:40
You may expect bonds between two atoms which each have n covalent lonic metallic hydrogen
Answers: 2

Chemistry, 21.06.2019 22:30
Explain why scientists use shared characteristics to make cladograms.
Answers: 1

Chemistry, 22.06.2019 05:10
How many miles of water are produced if 5.43 mol pbo2 are consumed
Answers: 1

Chemistry, 22.06.2019 07:00
In the cathode ray tube experiment, j. j. thomson passed an electric current through different gases inside a cathode ray tube in the presence of an electric field. in which two ways did this experiment change scientists’ understanding of the atom?
Answers: 2
You know the right answer?
The picture shows two containers filled with a gas, both initially at room temperature.
A Heat
Questions

Mathematics, 09.07.2021 07:20

Mathematics, 09.07.2021 07:20


History, 09.07.2021 07:20

Biology, 09.07.2021 07:20

Mathematics, 09.07.2021 07:20

Health, 09.07.2021 07:20

Mathematics, 09.07.2021 07:20


Social Studies, 09.07.2021 07:20

Physics, 09.07.2021 07:20



Mathematics, 09.07.2021 07:20

English, 09.07.2021 07:20

Mathematics, 09.07.2021 07:20

Spanish, 09.07.2021 07:20

Mathematics, 09.07.2021 07:20

Mathematics, 09.07.2021 07:20



