Chemistry, 24.05.2021 09:10 alexkrol30083
A 5.00 mL sample of hydrochloric acid is titrated with 0.1293 M ammonia (a base). If the titration required 28.15 mL of ammonia, determine the following:
the original concentration of the acid
the original pH of the acid

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 17:40
If 3 moles of a compound use 24 j of energy in a reaction, what is the a hreaction in j/mol?
Answers: 1

Chemistry, 22.06.2019 18:40
What is the binding energy of a nucleus that has a mass defect of 5.81*10-^29 kg a 5.23*10-^12 j b 3.15* 10^12 j c 1.57*10-3 j d 9.44*10^20 j
Answers: 1

Chemistry, 22.06.2019 23:30
How many grams of ammonia would be produced by the decomposition of 16.93 mlof hydrazine? (the density of hydrazine is 1.021g/ml)
Answers: 3

Chemistry, 23.06.2019 03:30
27 drag each label to the correct location on the image. a particular exosolar system has five planets in total: a, b, c, d, and e. the table lists the orbital periods of these planets in days. planet orbital period (days) a 600 b 80 c 1,000 d 500 e 100 move each planet to its orbit in the system.
Answers: 3
You know the right answer?
A 5.00 mL sample of hydrochloric acid is titrated with 0.1293 M ammonia (a base). If the titration r...
Questions

Chemistry, 15.12.2020 21:30



Physics, 15.12.2020 21:30

Mathematics, 15.12.2020 21:30




Social Studies, 15.12.2020 21:30

English, 15.12.2020 21:30



Mathematics, 15.12.2020 21:30



Spanish, 15.12.2020 21:30

Mathematics, 15.12.2020 21:30


Mathematics, 15.12.2020 21:30

Mathematics, 15.12.2020 21:30

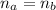

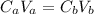
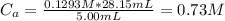
![pH = -log([H^{+}])](/tpl/images/1343/3808/0d4b9.png)