
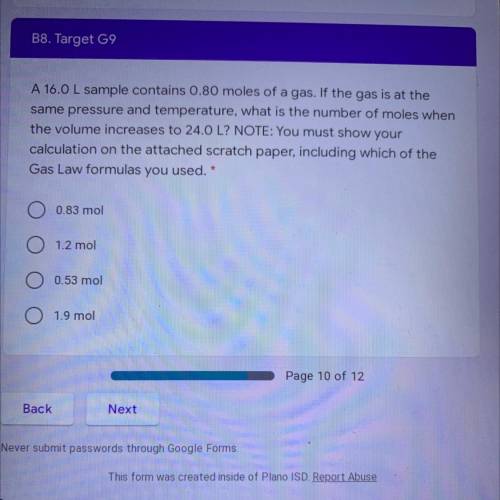
A 16.0 L sample contains 0.80 moles of a gas. If the gas is at the
same pressure and temperature, what is the number of moles when
the volume increases to 24.0 L? NOTE: You must show your
calculation on the attached scratch paper, including which of the
Gas Law formulas you used. *
A. 0.83 mol
B. 1.2 mol
C. 0.53 mol
D. 1.9 mol
(Please show your work)

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:00
In an investigation that uses the scientific method, which step immediately follows making a hypothesis? o summarizing the results o asking a question o making observations designing an experiment mark this and retum save and exit next submit
Answers: 2

Chemistry, 22.06.2019 20:10
Suppose you mix one mole of sulfuric acid (h2so4) with 1 mole of sodium hydroxide(naoh). why does the ph of the solution remain below 7? ( explain so i can get better understanding! )
Answers: 2

Chemistry, 23.06.2019 01:00
What type of chemical bond is formed between two atoms of bromine 1. metallic 2. hydrogen 3. ionic 4. covalent
Answers: 1

Chemistry, 23.06.2019 01:30
If a particle has z = 25 and 23 electrons, what is its charge?
Answers: 2
You know the right answer?
A 16.0 L sample contains 0.80 moles of a gas. If the gas is at the
same pressure and temperature, w...
Questions


Mathematics, 16.07.2019 14:00


Biology, 16.07.2019 14:00




Geography, 16.07.2019 14:00


Social Studies, 16.07.2019 14:00

English, 16.07.2019 14:00

Chemistry, 16.07.2019 14:00

Mathematics, 16.07.2019 14:00

Mathematics, 16.07.2019 14:00

Social Studies, 16.07.2019 14:00




Biology, 16.07.2019 14:00

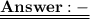
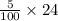 = 120/100= 12/10= 1.2 Mole ( Ans )
= 120/100= 12/10= 1.2 Mole ( Ans )

