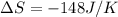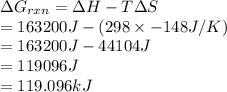AHM

Chemistry, 22.05.2021 20:00 bankscorneliuso39
Calculate AGrxn for this equation, rounding your
answer to the nearest whole number.
AHM
2N2(g) + O2(g) → 2N (9)
163.2 kJ
ASX = -148 J/K
T = 298 K
AGO
kJ

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 13:00
Dwayne filled a small balloon with air at 298.5 k. he put the balloon into a bucket of water, and the water level in the bucket increased by 0.54 liter. if dwayne puts the balloon into a bucket of ice water at 273.15 k and waits for the air inside the balloon come to the same temperature, what will the volume of the balloon be? assume the pressure inside the balloon doesn’t change. type the correct answer in the box. express your answer to the correct number of significant figures. the volume of the balloon at 273.15 k is liters.
Answers: 2

Chemistry, 21.06.2019 17:00
What is the empirical formula of vanadium 1 oxide given that 20.38 grams of vandium combines with oxygen to form 23.58 grams of the oxide
Answers: 1

Chemistry, 22.06.2019 08:30
In a chemical reaction at equilibrium, the rate of the forward reaction the rate of the reverse reaction. if the rate of the forward reaction more products are formed.
Answers: 1

You know the right answer?
Calculate AGrxn for this equation, rounding your
answer to the nearest whole number.
AHM
AHM
Questions



History, 13.11.2020 21:00




Social Studies, 13.11.2020 21:00


History, 13.11.2020 21:00

Mathematics, 13.11.2020 21:00

Social Studies, 13.11.2020 21:00

Chemistry, 13.11.2020 21:00


History, 13.11.2020 21:00

Mathematics, 13.11.2020 21:00

Biology, 13.11.2020 21:00




Mathematics, 13.11.2020 21:00

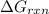 for given equation is 119.096 kJ.
for given equation is 119.096 kJ. 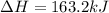 (1 kJ = 1000 J) = 163200 J
(1 kJ = 1000 J) = 163200 J