
Chemistry, 21.05.2021 20:10 dari122223
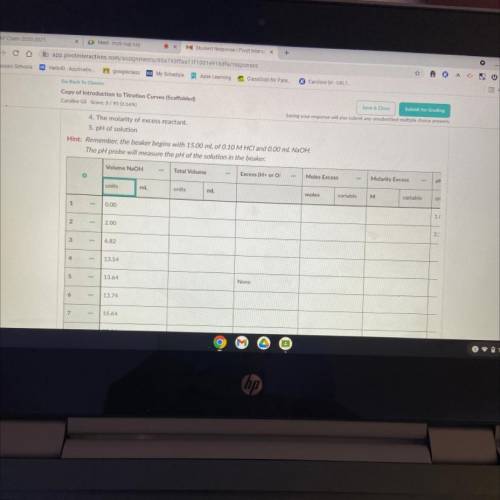
6. Now that you've predicted the equivalence point, let's predict several other points on the titration curve.
Perform calculations to predict the pH at the following points in the titration.
• 0.00 ml NaOH added
• 2.00 ml NaOH
• Half of the equivalence point volume
• 0.10 ml before the equivalence point
• Equivalence Point
• 0.10 mL after equivalence point
• 2.00 mL after the equivalence point
• 10.00 mL after equivalence point

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:00
Temperature and kinetic energy are proportional. a) adirectly b) directly c) indirectly
Answers: 2


Chemistry, 23.06.2019 00:30
Gasoline has a density of 0.740 g/ml. if you have 328 grams of gasoline, what is the volume in milliliters?
Answers: 1

You know the right answer?
6. Now that you've predicted the equivalence point, let's predict several other points on the titrat...
Questions


Geography, 08.09.2019 02:20



English, 08.09.2019 02:20


Mathematics, 08.09.2019 02:20


Mathematics, 08.09.2019 02:20






Health, 08.09.2019 02:20

Health, 08.09.2019 02:20



Health, 08.09.2019 02:20

Health, 08.09.2019 02:20



