
Chemistry, 20.05.2021 18:40 Chewychipsx4023
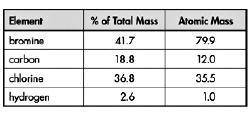
In order to identify an unknown compound found at an environmental cleanup site, an analytical chemist determines the percent composition of the compound. The results are shown in the table below.
What is the empirical formula of the compound?
a
C3H5Cl2Br
b
CHClBr
c
C4HCl5Br10
d
C3H5ClBr2

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 20:30
The balanced chemical equation for this lab is: 3cucl2(aq) + 2al(s) 3cu(s) + 2alcl3(aq) if 10.5 g copper chloride react with 12.4 g aluminum, what is the limiting reactant?
Answers: 3


Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

You know the right answer?
In order to identify an unknown compound found at an environmental cleanup site, an analytical chemi...
Questions



English, 19.11.2020 21:00

Mathematics, 19.11.2020 21:00

Mathematics, 19.11.2020 21:00

Mathematics, 19.11.2020 21:00

Biology, 19.11.2020 21:00




Mathematics, 19.11.2020 21:00

English, 19.11.2020 21:00


Mathematics, 19.11.2020 21:00






English, 19.11.2020 21:00



