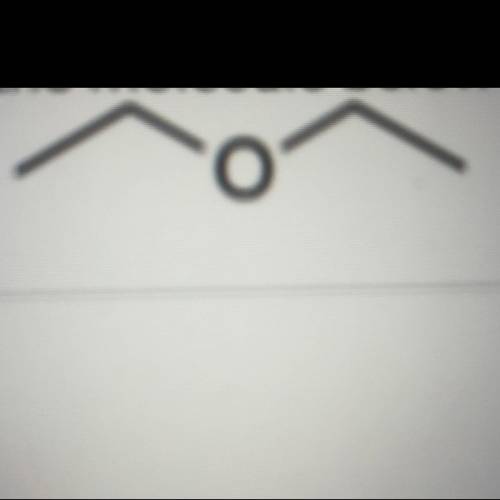
According to arrhenius theory of acid and bases, an acid is a proton donor, while base donates hydroxyl ions. further, acid and base reacts to form salt and water.consider following example.hcl + h2o → cl- + h3o+here, hcl has donated a proton, hence it is an acid.now consider following reactionhcl + naoh ↔ nacl + h2oin this reaction, hcl donates proton, hence it is acid. on other hand, naoh donates hydroxyl ion (i.e. hence it is a base.