
Chemistry, 18.05.2021 19:30 kmontello5
Equal molar quantities of Ca2 and EDTA (H4Y) are added to make a 0.010 M solution of CaY2- at pH 10. The formation constant for CaY2- is 5.0 x 1010 and the fraction of unprotonated EDTA (Y4-) is 0.35 at pH10. Calculate the concentration of free Ca2 in this solution.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 14:40
Choose an equation that represents an enzyme-catalyzed reaction. (a) enzyme + substrate → enzyme-substrate complex (b) enzyme + substrate ←−→ enzyme + products (c) enzyme + substrate ←−→ enzyme-substrate complex → enzyme + products (d) enzyme + substrate ←−→ enzyme-substrate complex → enzyme-substrate complex + products
Answers: 2

Chemistry, 22.06.2019 23:30
Rank substituents in order of their priority when assigning the e or z label to an alkene. i, ch2i , h, ch2ch2cl, f
Answers: 2

Chemistry, 23.06.2019 03:00
Achemical equilibrium between gaseous reactants and products is shown. n2(g) + 3h2(g) ⇌ 2nh3(g) how will the reaction be affected if the pressure on the system is increased? it will shift toward the reactant side as there is lower pressure on the reactant side. it will shift toward the product side as there is higher pressure on the product side. it will shift toward the reactant side as there are a greater number of moles of gas on the reactant side. it will shift toward the product side as there are a fewer number of moles of gas on the product side.
Answers: 2

Chemistry, 23.06.2019 03:30
How many grams of sodium chloride are in 250ml of a 2.5m naci solution
Answers: 1
You know the right answer?
Equal molar quantities of Ca2 and EDTA (H4Y) are added to make a 0.010 M solution of CaY2- at pH 10....
Questions


Health, 27.11.2020 21:20

Mathematics, 27.11.2020 21:20

Advanced Placement (AP), 27.11.2020 21:20

Arts, 27.11.2020 21:20




Biology, 27.11.2020 21:20


Spanish, 27.11.2020 21:20

Mathematics, 27.11.2020 21:20




Mathematics, 27.11.2020 21:20


Chemistry, 27.11.2020 21:20

Mathematics, 27.11.2020 21:20

Chemistry, 27.11.2020 21:20

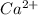 +
+ 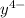 ⇄
⇄ 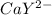
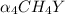 ; ∝₄ = 0.35
; ∝₄ = 0.35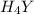 ⇄
⇄ 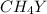 =
= 

