
Chemistry, 18.05.2021 19:20 catzdatbloadd
It is desired that an acetic acid sodium acetate buffered solution have a pH of 5.27. You have a solution that has an acetic acid concentration of 0.01 M. What molarity of sodium acetate will you need to add to the solution, given that the pKa of acetic acid is 4.74

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Which of the following ocean acidification? are the most likely side effects of a ph less than 7.0 in the ocean b. more metal salts altering the ocean chemistry c. dissolution of the shells of marine organisms d. both a & b e. all of the above.
Answers: 3



Chemistry, 23.06.2019 04:20
The reaction below shows a system in equilibrium. how would a decrease in temperature affect this reaction? a. the rate of formation of the gases would increase. b. the equilibrium of the reaction would shift to the left. c. the equilibrium would shift to cause the gases to sublime into solids. d. the chemicals on the left would quickly form the chemical on the right.
Answers: 1
You know the right answer?
It is desired that an acetic acid sodium acetate buffered solution have a pH of 5.27. You have a sol...
Questions

Mathematics, 27.05.2021 22:40

Mathematics, 27.05.2021 22:40


Mathematics, 27.05.2021 22:40


Mathematics, 27.05.2021 22:40

Mathematics, 27.05.2021 22:40



Mathematics, 27.05.2021 22:40


Mathematics, 27.05.2021 22:40

Mathematics, 27.05.2021 22:40




Mathematics, 27.05.2021 22:40

Mathematics, 27.05.2021 22:40


History, 27.05.2021 22:40

![pH = pK_a + \log \frac{[A^-]}{[HA]}](/tpl/images/1331/4715/1110c.png)
![[Ac^-]](/tpl/images/1331/4715/fda84.png) = Concentration of the conjugate base
= Concentration of the conjugate base 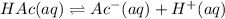
![[Ac^-] = ?](/tpl/images/1331/4715/57252.png)
![5.27= 4.74 + \log \frac{[Ac^-]}{[0.01 M]}](/tpl/images/1331/4715/91873.png)
![[Ac^-]=0.0339 M\approx 0.034M](/tpl/images/1331/4715/e0d23.png)
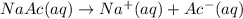
![[Ac^-]=[Na^+]=[NaAc]= 0.034M](/tpl/images/1331/4715/0e140.png)


