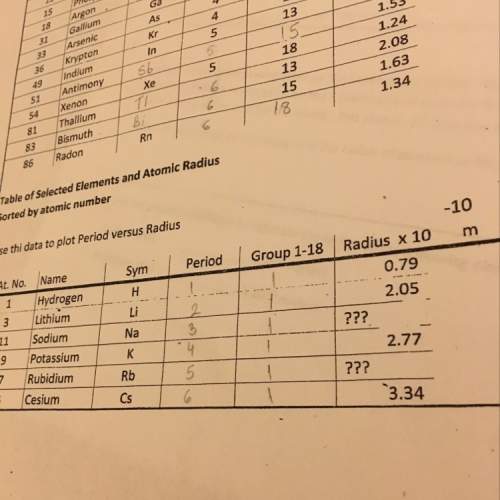
Chemistry, 18.05.2021 19:20 shataviasumpter78
The atomic radius of chlorine is 102 pm. Explain why the radius of Cl in the perchlorate anion is 41 pm, whereas the radius of Cl in the chloride anion is 181 pm. Compare both values to the atomic radius.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:30
Sarah wants to know where in her garden chamomile would grow the best. she thinks chamomile will grow best in the corner of the garden that gets the most sunlight. to test her hypothesis, she decides to plant several groups of chamomile in her garden as an experiment. which of the following variables will sarah need to measure to know which group of plants grew best? a. the location of the plants b. the type of plants c. the height of the plants d. the amount of water she gives the plants
Answers: 1

Chemistry, 22.06.2019 03:00
What happened in 2012 and how does it illustrate the importance of understanding the sun and how it works?
Answers: 3


Chemistry, 22.06.2019 18:00
Many pharmaceutical drugs are organic compounds that were originally synthesized in the laboratory. which two scientific disciplines are bridged by pharmaceutical drugs? chemistry and forensics chemistry and medicine biology and forensics biology and criminology
Answers: 2
You know the right answer?
The atomic radius of chlorine is 102 pm. Explain why the radius of Cl in the perchlorate anion is 41...
Questions

Mathematics, 23.10.2020 04:01

Mathematics, 23.10.2020 04:01

English, 23.10.2020 04:01

Mathematics, 23.10.2020 04:01


Mathematics, 23.10.2020 04:01



Mathematics, 23.10.2020 04:01

Mathematics, 23.10.2020 04:01

English, 23.10.2020 04:01


Mathematics, 23.10.2020 04:01



History, 23.10.2020 04:01

History, 23.10.2020 04:01


Chemistry, 23.10.2020 04:01

Mathematics, 23.10.2020 04:01