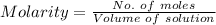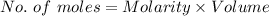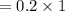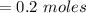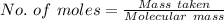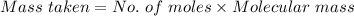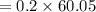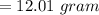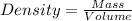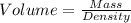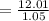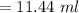Chemistry, 18.05.2021 19:10 phancharamachasm
You must make 1 L of 0.2 M acetic acid (CH3COOH). All you have available is concentrated glacial acetic acid (assay value, 98%; specific gravity, 1.05 g/mL). It will take milliliters of acetic acid to make this solution. Assume a gram molecular weight of 60.05 grams.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 12:30
Which statement is true about this reaction? 14n+1h 15o it is a practical source of energy on earth. it occurs only outside the solar system. its product is heavier than each of its reactants. it shows the critical mass of an element.
Answers: 2

Chemistry, 22.06.2019 19:00
How many liters of ethylene glycol antifreeze (c2h6o2), with a density of 1.100 g/l, would you add to your car radiator containing 15.0 kg of water if you needed to protect your engine to - 21.5°c? for water, kf = 1.86°c m -1.
Answers: 1

Chemistry, 22.06.2019 22:00
Scientists often have to deal with numbers that are either very large or very small. for example, the radius of the sun is approximately 696,000 kilometers, while bacterial cells are as small as 1.9 × 10-4 millimeters. express each number in an alternate form.
Answers: 1
You know the right answer?
You must make 1 L of 0.2 M acetic acid (CH3COOH). All you have available is concentrated glacial ace...
Questions



Computers and Technology, 18.07.2020 02:01












Spanish, 18.07.2020 02:01

Mathematics, 18.07.2020 02:01




English, 18.07.2020 02:01