1. Using the following BALANCED chemical equation.
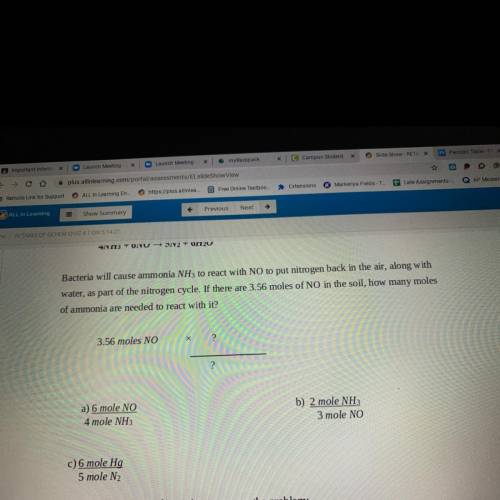
4NH3 + 6NO5N2 + 6H2O
Bacteria will cause...

Chemistry, 18.05.2021 17:50 kraigstlistt
1. Using the following BALANCED chemical equation.
4NH3 + 6NO5N2 + 6H2O
Bacteria will cause ammonia NH3 to react with NO to put nitrogen back in the air, along with
water, as part of the nitrogen cycle. If there are 3.56 moles of NO in the soil, how many moles
of ammonia are needed to react with it?
3.56 moles NO
?
?
a) 6 mole NO
4 mole NH3
b) 2 mole NH:
3 mole NO
c) 6 mole Hg
5 mole N

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 13:00
What type of reaction is represented by the following example? 2co2 (g) + 4h2o (l) + 1452 kj 2ch3oh (l) (g) + 3o2 (g) exothermic endothermic
Answers: 1

Chemistry, 22.06.2019 08:00
An electron moved from shell n = 2 to shell n = 1. what most likely happened during the transition? a fraction of a photon was added. a photon of energy was absorbed. a fraction of a photon was removed. a photon of energy was released.
Answers: 1


Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3
You know the right answer?
Questions

Chemistry, 16.10.2021 21:00




Physics, 16.10.2021 21:00



History, 16.10.2021 21:00

Mathematics, 16.10.2021 21:00

Chemistry, 16.10.2021 21:00

Mathematics, 16.10.2021 21:00


Mathematics, 16.10.2021 21:00


Mathematics, 16.10.2021 21:00



History, 16.10.2021 21:00

Mathematics, 16.10.2021 21:00

Mathematics, 16.10.2021 21:00



