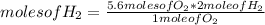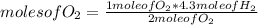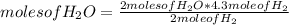Chemistry, 17.05.2021 16:50 genyjoannerubiera
How many moles of water can be produced with 4.3 moles of H2 and 5.6 moles of O2? Which reactant is limiting? How many moles of the excess reactant will be left after the reaction? 2 H2 + O2 2 H2O

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 14:00
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3

Chemistry, 23.06.2019 07:00
What is the difference between covalent bonds and ionic bonds? covalent bonds involve the sharing of electrons between atoms; ionic bonds involve the electrical attraction between charged atoms. covalent bonds involve the transfer of electrons between charged atoms; ionic bonds involve the sharing of electrons between atoms. covalent bonds involve the sharing of pairs of electrons between atoms; ionic bonds involve the sharing of single electrons between atoms. covalent bonds involve the sharing of electrons between atoms; ionic bonds involve the sharing of protons between charged atoms.
Answers: 1

Chemistry, 23.06.2019 07:10
1) a light bulb takes in 30 of energy per second. it transfers 3j as use energy. calculate the efficiency. second. it transfers 3j as useful light energy and 27j as heat energy. calculate the efficiency
Answers: 1
You know the right answer?
How many moles of water can be produced with 4.3 moles of H2 and 5.6 moles of O2? Which reactant is...
Questions



Mathematics, 27.10.2020 02:10



English, 27.10.2020 02:10

History, 27.10.2020 02:10



Mathematics, 27.10.2020 02:10






Social Studies, 27.10.2020 02:10


History, 27.10.2020 02:10

Physics, 27.10.2020 02:10