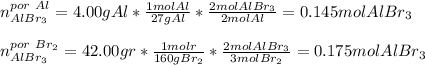Chemistry, 15.05.2021 06:10 dropnsocks8315
Se hace reaccionar 4,00 g de aluminio y 42,00 g de bromo, según la reacción: Al(s)+Br2(l)⟶AlBr3(s) Calcular las moles de AlBr3(s) producido. PA: Al=27; Br=80

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:30
Both josef loschmidt and amedeo avogadro contributed to our understanding of basic molecular numbers, sizes, and reaction ratios. neither scientist discovered “avogadro’s number” in the form we use it today (6.02 x 10 23). still, there’s a controversy over the name. research the contributions from these two scientists and read about how avogadro’s number got its name. briefly state what you think this number should be called, providing key details of each scientist’s contributions to this concept and a solid rationale for your case in naming the number.
Answers: 2

Chemistry, 22.06.2019 08:30
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2

Chemistry, 22.06.2019 12:30
What metric units would you use to measure the thickness of a key
Answers: 3

Chemistry, 22.06.2019 14:30
The three types is stress that act on earths rocks are compression, tension, and
Answers: 1
You know the right answer?
Se hace reaccionar 4,00 g de aluminio y 42,00 g de bromo, según la reacción: Al(s)+Br2(l)⟶AlBr3(s) C...
Questions








History, 21.08.2019 17:30


English, 21.08.2019 17:30


Physics, 21.08.2019 17:30

English, 21.08.2019 17:30


History, 21.08.2019 17:30

Geography, 21.08.2019 17:30

History, 21.08.2019 17:30

German, 21.08.2019 17:30


Business, 21.08.2019 17:30