Name the functional group in the
following molecule:
HÖCH2CH2CH3
A, ketone
B, ald...

Chemistry, 15.05.2021 02:50 mariaramirez013
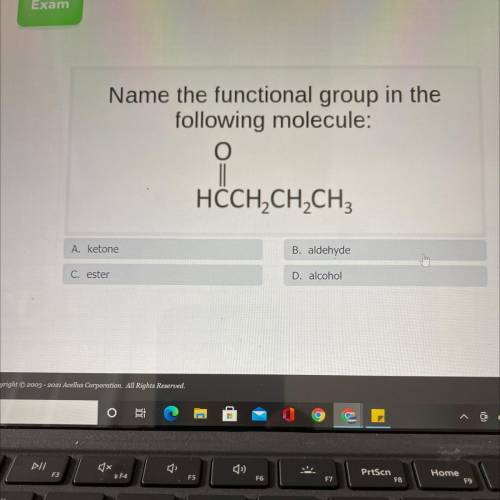
Name the functional group in the
following molecule:
HÖCH2CH2CH3
A, ketone
B, aldehyde
C. ester
D, alcohol

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 14:50
Your roll: experienced electron speech is adressed to: a new "freshman class" of electrons job: write a speech task: you are to pretend that you are giving a speech to a new group of electrons. be sure to mention their placement in an atom, their charge, and their role in chemical bonding (ionic and covalent) be specific!
Answers: 3

Chemistry, 22.06.2019 06:00
24. a sports ball is inflated to an internal pressure of 1.85 atm at room temperature (25 °c). if the ball is then played with outside where the temperature is 7.5 °c, what will be the new pressure of the ball? assume the ball does not change in volume nor does any air leak from the ball a) 0.555 atm b) 1.74 atm c) 1.85 atm d) 1.97 atm
Answers: 2

Chemistry, 22.06.2019 07:00
Indicate whether the specified alkyl halides will form primarily substitution products, only elimination products, both substitution and elimination products, or no products when they react with sodium methoxide. 1-bromobutane 1-bromo-2-methylpropane 2-bromobutane 2-bromo-2-methylpropane
Answers: 2

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3
You know the right answer?
Questions

Mathematics, 08.12.2020 19:50


Mathematics, 08.12.2020 19:50



Mathematics, 08.12.2020 19:50

Mathematics, 08.12.2020 19:50

History, 08.12.2020 19:50

Mathematics, 08.12.2020 19:50

Physics, 08.12.2020 19:50


Biology, 08.12.2020 19:50

Mathematics, 08.12.2020 19:50




Mathematics, 08.12.2020 19:50



Mathematics, 08.12.2020 19:50



