Answers: 2
Another question on Chemistry

Chemistry, 20.06.2019 18:02
Explain the effects of nh3 and hcl on the cuso4 solution in terms of le chatelier's principle
Answers: 2

Chemistry, 22.06.2019 06:00
If you burn 10 kilograms of wood in a fire (combustion) what is the weight of the products after the fire has finished burning the wood?
Answers: 3

Chemistry, 22.06.2019 09:00
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1

Chemistry, 22.06.2019 10:30
If you add 5.00 ml of 0.100 m sodium hydroxide to 50.0 ml of acetate buffer that is 0.100 m in both acetic acid and sodium acetate, what is the ph of the resulting solution? acetic acid: ka = 1.8. x 10-5
Answers: 1
You know the right answer?
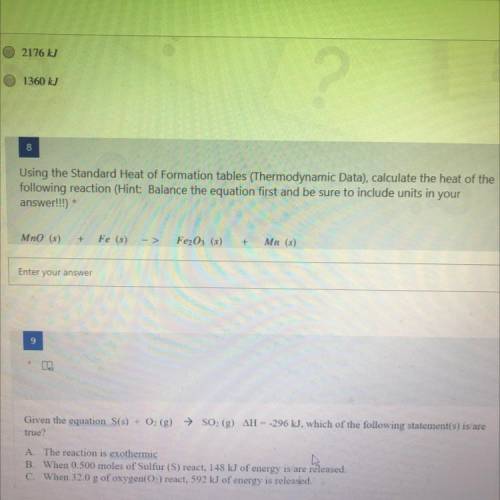
Using the Standard Heat of Formation tables (Thermodynamic Data), calculate the heat of the
followi...
Questions

Biology, 04.02.2021 01:00



Mathematics, 04.02.2021 01:00

Mathematics, 04.02.2021 01:00

Mathematics, 04.02.2021 01:00



Mathematics, 04.02.2021 01:00

Mathematics, 04.02.2021 01:00


Mathematics, 04.02.2021 01:00



English, 04.02.2021 01:00



Mathematics, 04.02.2021 01:00

Health, 04.02.2021 01:00

SAT, 04.02.2021 01:00