
Chemistry, 14.05.2021 23:20 saltyimps3
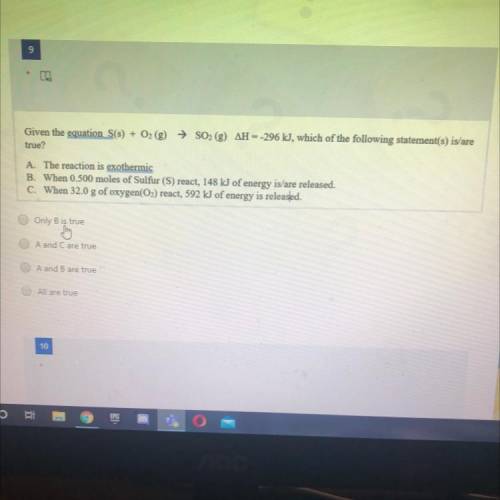
Given the equation S(s) + O2 (g) → SO2 (g) AH --296 kJ, which of the following statement(s) is/are
true?
A. The reaction is exothermic
B. When 0.500 moles of Sulfur (S) react, 148 kJ of energy is/are released.
C. When 32.0 g of oxygen(O2) react, 592 kJ of energy is released.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 09:00
Particles vibrate in a rigid structure and do not move relative to their neighbors.
Answers: 1

Chemistry, 22.06.2019 11:00
Iron (3) oxide will decompose in the presence of hydrogen gas and heater to produced iron and digydrogen monoxide white a balanced chemical equation
Answers: 1

Chemistry, 22.06.2019 19:30
What is the mass of oxygen gas is consumed in a reaction that produces 4.60mol so2
Answers: 3

You know the right answer?
Given the equation S(s) + O2 (g) → SO2 (g) AH --296 kJ, which of the following statement(s) is/are...
Questions




History, 25.06.2019 00:30


Biology, 25.06.2019 00:30


Mathematics, 25.06.2019 00:30





Mathematics, 25.06.2019 00:30






Mathematics, 25.06.2019 00:30



