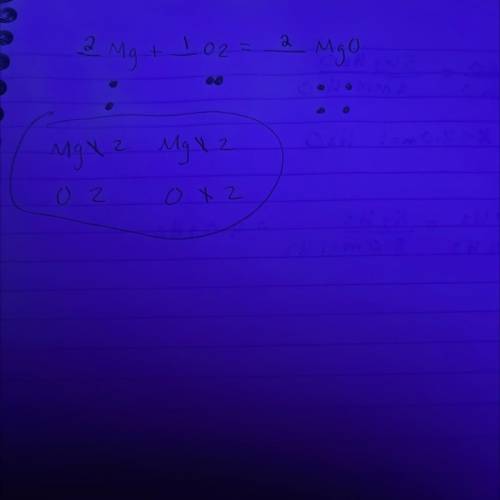In a balanced chemical reaction, the number of atoms of each element in the product(s) always equals the
A. molar mass of the reactants
B. proportional masses of the reactants
C. the number of atoms of each element in the reatants
D. the number of elements in the reactants

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 10:50
How many grams of oxygen gas are contained in a 15 l sample at 1.02 atm and 28°c? show your work.
Answers: 1

Chemistry, 22.06.2019 19:00
What information does a complete ionic equation give that the balanced equation doesn’t show?
Answers: 1

Chemistry, 22.06.2019 20:30
How many grams of phosphorus are contained in 5.09 moles of phosphorus?
Answers: 1

Chemistry, 22.06.2019 21:00
Rays from the sun are not considered matter true or false
Answers: 2
You know the right answer?
In a balanced chemical reaction, the number of atoms of each element in the product(s) always equals...
Questions

History, 25.07.2019 23:00

English, 25.07.2019 23:00

Mathematics, 25.07.2019 23:00

Chemistry, 25.07.2019 23:00

History, 25.07.2019 23:00




History, 25.07.2019 23:00

Mathematics, 25.07.2019 23:00


Biology, 25.07.2019 23:00


Social Studies, 25.07.2019 23:00


Biology, 25.07.2019 23:00

History, 25.07.2019 23:00


English, 25.07.2019 23:00

Mathematics, 25.07.2019 23:00