
Chemistry, 13.05.2021 23:00 honeylebling
Consider the chemical equation. 2NBr3 + 3NaOH Right arrow. N2 + 3NaBr + 3HOBr If there are 40 mol of NBr3 and 48 mol of NaOH, what is the excess reactant?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:40
Which statement is usually true about the relationship between activation energy and reaction rates? low activation energy barriers result in low rates. high activation energy barriers result in low rates. low activation energy barriers result in no reaction. high activation energy barriers result in no reaction.
Answers: 3

Chemistry, 23.06.2019 01:00
Who examines and coordinates the cleanup of polluted sites?
Answers: 2

Chemistry, 23.06.2019 04:20
The equation below shows the reaction of zinc with hydrochloric acid (hcl). zn (s) + 2 hcl (aq) —> zncl2 (aq) + h2 (g) what will happen if the concentration of hcl is decreased? a. more zncl2 will be produced. b. the reaction rate will slow down. c. the hydrochloric acid will become more acidic. d. the reaction will produce water instead of hydrogen gas.
Answers: 1

Chemistry, 23.06.2019 07:30
In the diagram here that represents the reaction, which reactant, a or b, is the limiting reagent?
Answers: 1
You know the right answer?
Consider the chemical equation. 2NBr3 + 3NaOH Right arrow. N2 + 3NaBr + 3HOBr If there are 40 mol of...
Questions

Mathematics, 08.12.2021 20:20

Mathematics, 08.12.2021 20:20

English, 08.12.2021 20:20





English, 08.12.2021 20:20

Mathematics, 08.12.2021 20:20

Mathematics, 08.12.2021 20:20

History, 08.12.2021 20:20

English, 08.12.2021 20:20

Geography, 08.12.2021 20:20



Mathematics, 08.12.2021 20:20

SAT, 08.12.2021 20:20



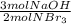 =60 moles NaOH
=60 moles NaOH

