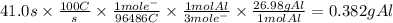Chemistry, 13.05.2021 20:50 naomicervero
In the Hall-Heroult process, a large electric current is passed through a solution of aluminum oxide Al2O3 dissolved in molten cryolite Na3AlF6, resulting in the reduction of the Al2O3 to pure aluminum. Suppose a current of 100.A is passed through a Hall-Heroult cell for 41.0 seconds. Calculate the mass of pure aluminum produced. Round your answer to 3 significant digits. Also, be sure your answer contains a unit symbol.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:20
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1

Chemistry, 22.06.2019 10:30
Geothermal energy for industrial use is available almost anywhere. a.true b.false
Answers: 2


Chemistry, 22.06.2019 21:00
Rays from the sun are not considered matter true or false
Answers: 2
You know the right answer?
In the Hall-Heroult process, a large electric current is passed through a solution of aluminum oxide...
Questions

Mathematics, 06.01.2020 05:31


Chemistry, 06.01.2020 05:31



History, 06.01.2020 05:31



Mathematics, 06.01.2020 05:31



Mathematics, 06.01.2020 05:31

Spanish, 06.01.2020 05:31





Geography, 06.01.2020 05:31

Chemistry, 06.01.2020 05:31

Mathematics, 06.01.2020 05:31