
Chemistry, 13.05.2021 04:00 catherinesquitieri
An atom has the following electron configuration 1s2s2sp63s23p64s2 How many valance electrons does this atom have?
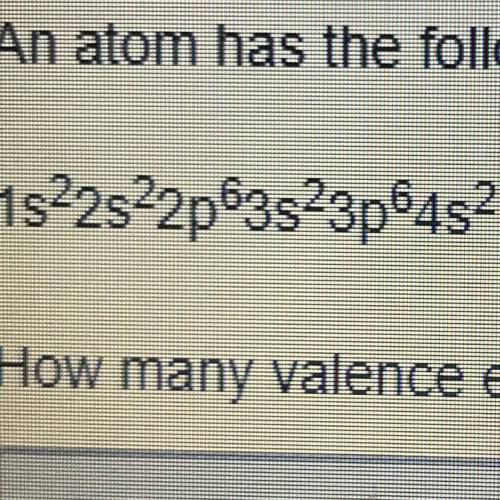

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 08:30
How would the number of moles (n) of o2 change if the atmospheric pressure doubled but all other variables stayed the same
Answers: 2

Chemistry, 22.06.2019 20:30
Draw a line graph showing the relationship between temperature in kelvin as a function of kinetic energy.
Answers: 3

Chemistry, 22.06.2019 23:30
The density of benzene at 15 °c is 0.8787 g/ml. calculate the mass of 0.1500 l of benzene at this temperature. enter your answer in terms of grams
Answers: 2

Chemistry, 23.06.2019 06:40
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants.thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction.ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch,are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1
You know the right answer?
An atom has the following electron configuration 1s2s2sp63s23p64s2 How many valance electrons does t...
Questions

Mathematics, 27.10.2020 04:10

Health, 27.10.2020 04:10

Mathematics, 27.10.2020 04:10


Chemistry, 27.10.2020 04:10



Mathematics, 27.10.2020 04:10


History, 27.10.2020 04:10


Mathematics, 27.10.2020 04:10

Arts, 27.10.2020 04:10

History, 27.10.2020 04:10

Mathematics, 27.10.2020 04:10



Mathematics, 27.10.2020 04:10

Physics, 27.10.2020 04:10



