Help!! I have a test
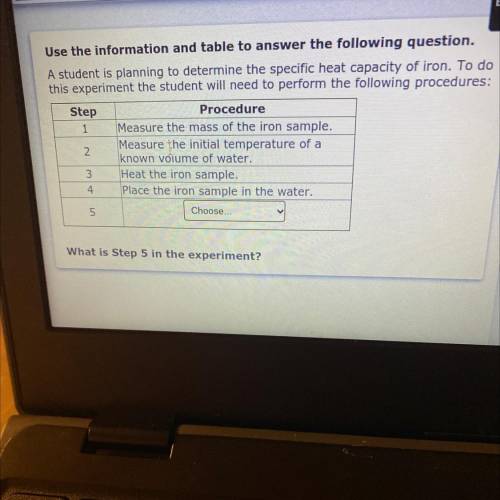
Use the information and table to answer the following question.
A studen...

Chemistry, 12.05.2021 16:20 michaelhal414
Help!! I have a test
Use the information and table to answer the following question.
A student is planning to determine the specific heat capacity of iron. To do
this experiment the student will need to perform the following procedures:
Step
Procedure
1 Measure the mass of the iron sample.
Measure the initial temperature of a
known volume of water.
3 Heat the iron sample.
4 Place the iron sample in the water.
N
.
5
Choose...
What is Step 5 in the experiment?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Determine the wavelength of the light absorbed when an electron in a hydrogen atom makes a transition from an orbital in the n=3 level to an orbital in the n=7 level.
Answers: 2

Chemistry, 22.06.2019 05:00
Frictional forces acting on an object are often converted into energy, which causes the temperature of the object to rise slightly.
Answers: 2

Chemistry, 22.06.2019 07:20
Part b: study of equilibrium on solubility: mg(oh)2(s) ⇌ mg2+(aq) + 2 oh–(aq) cloudy clear (pink) 7. a. b. 8. a. b. 9. 10. 11. 12. when adding concentrated hydrochloric acid, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 7a. you should indicate which ion was added to or removed from the equilibrium mixture. when adding edta, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 8a. you should indicate which ion was added to or removed from the equilibrium mixture. upon heating in which direction is the equilibrium shifting? upon cooling in which direction is the equilibrium shifting? is the forward reaction a. endothermic explain your answers to questions 9, 10, and 11. (l) left (r) right (l) left (r) right b. exothermic
Answers: 1

Chemistry, 22.06.2019 19:30
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2
You know the right answer?
Questions

Chemistry, 26.11.2021 21:40


English, 26.11.2021 21:40

Mathematics, 26.11.2021 21:50



History, 26.11.2021 21:50




Social Studies, 26.11.2021 21:50

Biology, 26.11.2021 21:50

Business, 26.11.2021 21:50

Social Studies, 26.11.2021 21:50



Social Studies, 26.11.2021 21:50

Chemistry, 26.11.2021 21:50

English, 26.11.2021 21:50

Chemistry, 26.11.2021 21:50



